Bayer to end sales of Essure, birth control device blamed for serious injuries
Bayer is voluntarily stopping sales of its controversial Essure sterilization device at the end of the year. Women who have been pushing to get it off the market are elated.
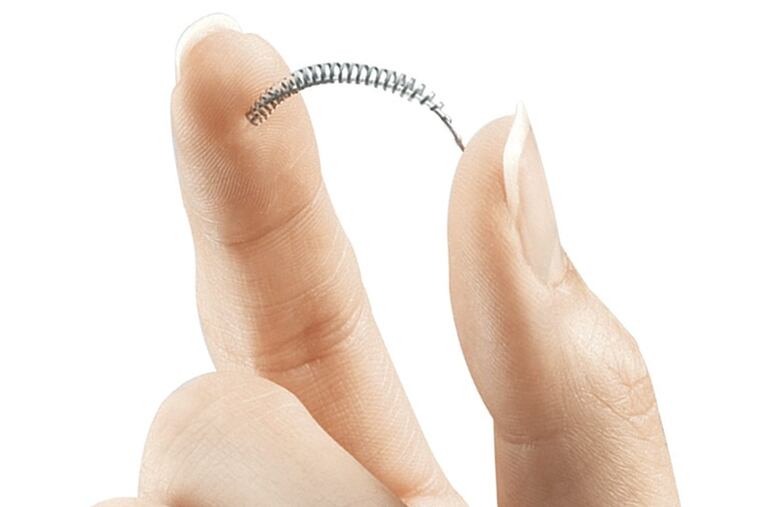
Pharmaceutical powerhouse Bayer on Friday announced that it will voluntarily end sales of its controversial Essure sterilization coils in the United States at the end of the year, giving a huge victory to women who blame the device for chronic pain, uterine perforations, miscarriages, hysterectomies, and other problems.
"I'm a bit in shock," said Angie Marie Firmalino, 45, of Tannersville, N.Y., a cofounder of the 37,000-member Essure Problems Facebook group. "This is what we've been fighting for for seven years. This is the last place in the world where Essure was still sold. No more women are going to be harmed by this device."
In a statement, Bayer said it continued to "stand behind the product's safety and efficacy," but was discontinuing distribution because recent declines in sales meant "the Essure business is no longer sustainable."
Essure was touted as a breakthrough alternative to surgical sterilization when it was approved by the U.S. Food and Drug Administration 16 years ago. It consists of two-inch long coils that are implanted in the fallopian tubes during a minimally invasive procedure. Scar tissue gradually forms, closing the tubes.
The device was developed by Conceptus Inc. and acquired by Bayer in 2013.
Bayer has said it doesn't know how many women have the implants, just that about a million Essure "kits" have been sold worldwide, most in the U.S.
In recent years, the ranks of women who claim harm from the device have ballooned, and more than 15,000 have filed "adverse event" reports with the FDA.
>> READ MORE: Essure leads to more repeat operations than tubal sterilization, study finds
In 2016, the FDA ordered Bayer to add label warnings that Essure can cause persistent pain, organ perforation, and allergic reactions. It also ordered the company to give doctors a three-page "patient decision checklist" to inform women about the risks.
More than 16,000 Essure users have filed lawsuits against Bayer. The company said it has gotten some thrown out, as well as narrowing the claims of the remaining suits.
On Tuesday, Firmalino and other Essure activists held a daylong protest outside Bayer's U.S. headquarters in Whippany, N.J. As part of it, they broadcast a clip from the forthcoming Netflix documentary about medical device failures called The Bleeding Edge. The film includes the stories of some women who claim they suffered permanent, serious complications from Essure.