5 questions: Breakthroughs in treatment for a common cause of vision loss among U.S. seniors
The standard treatment has been regular injections to the eyeball. So, not surprisingly, an active area of research has focused on finding alternatives, including promising gene therapy.
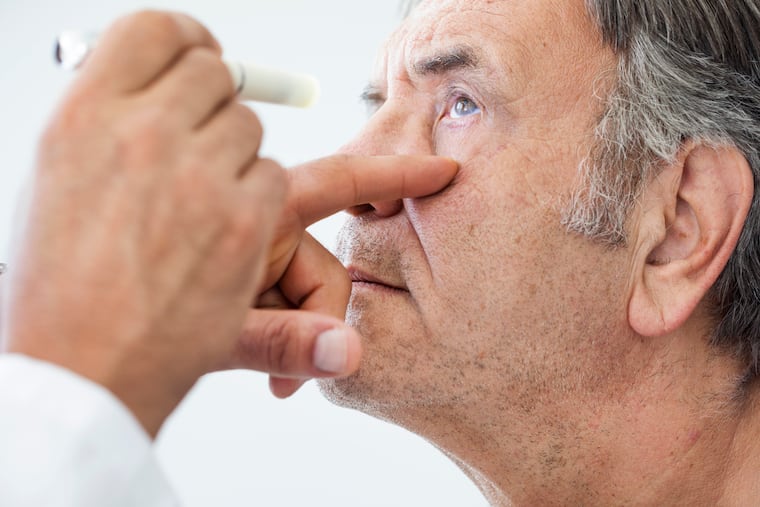
It might start with straight lines looking wavy. Or there might be a dark spot in the center of the person's vision.
A likely culprit: wet age-related macular degeneration, or wet AMD.
The standard treatment has been regular injections to the eyeball. So, perhaps it's not surprising that an active area of research has focused on finding alternatives, including promising gene therapy.
One of those researchers is Wills Eye Hospital retina specialist Allen Ho, who is the principal investigator of several clinical trials assessing new treatments for wet AMD. One of those trials is a gene-therapy treatment from Rockville, Md., biotechnology company Regenxbio, on whose scientific advisory board Ho serves.
Ho spoke to us recently about the disease, the treatments, and what's on the horizon.
What is wet AMD?
It is a common cause of vision loss among senior Americans. It affects the focus point of the eye, or the macula. The retina is like the film in the camera, and the macula is the focus point. It is an aging condition. There are two types – a dry form, and a wet form. Both can cause vision loss. But the wet form is typically the form that causes sudden changes in vision – sudden blurring, sudden loss of central vision. As its name implies, the wet form denotes leakage or bleeding in the macula.
Who develops wet AMD?
It affects a significant proportion of our seniors. The average age of the patient who develops wet AMD is in their 70s. Five percent to 10 percent of people in their 80s and 90s have either the wet or dry form. The dry form is much more prevalent. The dry form can be associated with central vision loss, but most people maintain reading and driving vision. What's exciting about the wet form is that we have treatments for it.
As for risk factors, age, obviously, is one. Also, genetics. But it's important to note that just because a family member has AMD, that does not mean it is your destiny.
Smoking is a significant risk factor. As for things you can do to reduce your risk: healthy habits; blood-pressure control; regular exercise, like walking 30 minutes a day. And a healthy diet. That includes eating a lot of different colors of fruits and vegetables, plus natural sources of omega-3 fatty acids, like salmon and tuna.
What is the current treatment for wet AMD?
Frequent eye injections, as often as monthly. This sounds intimidating. But it's an office procedure. We can anesthetize and sterilize the eyes. Many patients are very much relieved after they get their first injection because it's not as traumatic as you might imagine.
The substance we inject is to control the leakage. It's very effective in stabilizing vision. And sometimes we can improve vision, but not always. The key to the treatment of wet AMD is to catch it early. And the way to do that is to have an eye examination promptly when new visual symptoms – blurring, distortion or darkness — appear.
Although the current treatments are effective for the majority of patients, one of the problems is that patients typically have to come in every one to two months for injections. Sometimes, their adult child has to take a half-day off from work to bring them in. It can be a big deal.
But patients fear blindness and value vision. So they are willing to do this. If you're a grandparent and you're retired, you want to be independent and be able to read and drive your car and see your grandchildren's faces. This disease undermines that. So even though there's a burden of treatment — coming into the office every one to two months — those afflicted with wet AMD will do it.
Tell us about the new treatment you’re evaluating.
At Wills Eye, we are working on different types of longer-acting treatments, ones that may require less frequent visits to the eye-care team while still saving vision.
Gene therapy is a very exciting field now because there is a commercially approved gene-therapy product for another condition, another much less common retinal degeneration called Leber congenital amaurosis, or LCA. In this case, a defective gene is causing the vision loss. It prevents cells from making a specific protein required for vision. The first cradle of gene therapy for eye diseases is Philadelphia. The company that created the gene therapy for LCA is Spark Therapeutics of West Philadelphia.
In the Spark treatments, retina specialists essentially replace the defective gene with a good gene. They put the gene that works inside a non-disease-causing viral vector. The viruses are in a fluid, which is surgically delivered under the retina. You know what viruses do: They attach to your cells. If they're disease-causing, they might cause the common cold. But with these non-disease-causing viruses, they simply transport the gene to the defective cell. So now your cell can make the protein that is essential to the visual cycle. The science is pretty amazing.
That's the FDA-approved gene therapy. There's another way gene therapy can work. It can not only replace a defective gene — a mutation causing vision loss — but also can be used to introduce a gene that encodes for a therapeutic product similar to our current wet AMD injection medicines. That is what we are doing with Regenxbio.
The type of medicine that we're using now in the regular injections is called anti-VEGF. It stands for vascular endothelial growth factor, and it has miraculously transformed the outlook for wet AMD. But instead of patients coming in every month for an injection, what if we could use gene therapy to instruct the patient's own cells to produce the anti-VEGF medicine for the macula? And with gene therapy, it could last essentially indefinitely.
We're trying to create a sustainable drug-therapy system within the patient's own eyeball. Think about it: a sustainable one-procedure drug factory for your own eye disease.
This is what's exciting about the possibilities for gene therapy. Recently, at the annual meeting of the American Academy of Ophthalmology, our study team reported dose-dependent protein/drug production out to six months. Some of the study subjects no longer required their usual injections. That is exciting news.
How soon might this new treatment be available?
We are involved in the clinical trials at Wills Eye Hospital. We are looking at Phase II clinical trials in 2019. And, if they are positive, pivotal Phase III trials will follow in 2020. That's kind of appropriate, right? As in 20/20. We hope it could be on the market by 2023.
The real excitement in all this is for the patients. For someone coming in for monthly injections, this one-and-done treatment would be a sea change. The excitement for scientists and surgeons is that the science at Regenxbio and in gene therapy in general and the possibilities to help patients are incredible. With gene therapy, we're not talking about something that is pie in the sky. This is real. The concept has already been established. We still need to do more work, but the outlook is promising.