Coronavirus blood test to look for immunity gets a tryout in Chester County
While the approach is promising, the accuracy of such serology testing is unclear. Numerous countries have invested in tests that turned out to be unreliable

Chester County is at the forefront of a new approach to managing the COVID-19 pandemic: testing essential workers’ blood to see if they have developed immunity to the coronavirus, a sign they can safely go back to their jobs.
County officials announced Monday that they had purchased test kits from local firm Advaite, which promises to detect disease-fighting antibodies to the coronavirus. Testing will be launched this week, starting with workers from the county prison, a long-term health-care facility, and the county youth center. A system for testing first responders is in the works.
While the approach is promising, Renee Cassidy, public health physician with the Chester County Health Department, readily acknowledged that the accuracy of such serology testing is unclear. What’s more, she noted, numerous countries have invested in tests that turned out to be unreliable.
“Serology testing is very much in its infancy" for the coronavirus, Cassidy said. “It’s a little experimental.”
In theory, serology tests — so called because they examine the blood serum — could not only identify people who could safely go back to work, but also could answer questions about the transmissibility and lethality of the virus. Experts hope serology tests can play a role in diagnosing COVID-19 and even in treating critically ill patients. The idea, not yet proved, is that antibody-laden plasma from recovered patients could be given as a therapy.
Unlike the laborious, time-consuming molecular tests used to diagnose the coronavirus, serology tests can be done in clinics or doctor’s offices and provide results in less than an hour.
Because of the potential benefits, the U.S. Food and Drug Administration is allowing serology tests to come to market without review or emergency authorization. However, the tests may be used only to measure antibodies, not as the sole diagnostic test for sick patients. Some companies are seeking the FDA’s blessing, though they don’t have to; the first emergency authorization for a serology test was granted last week to North Carolina-based Cellex.









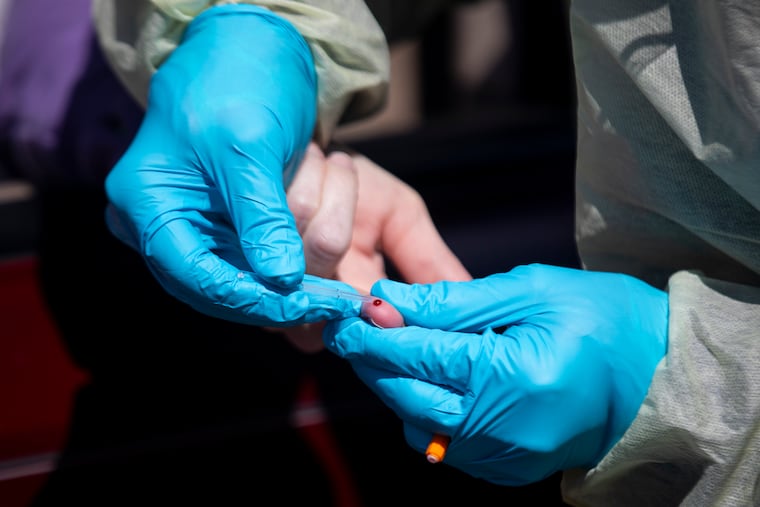


Advaite is also seeking FDA emergency authorization, but in news releases, the firm says its product is for rapid diagnosis — a use the FDA explicitly forbids. The company did not respond to a request for clarification.
In Chester County, where there have been 304 confirmed cases and three deaths, serology will not be used for diagnostic purposes.
“This test is supplemental to the coronavirus testing that we have already been doing and will continue to do,” said Jeanne Casner, Chester County Health Department director. “It is not a replacement test for confirming cases. We are undertaking the antibody blood test as another weapon in the fight to control coronavirus. Knowledge of who has developed antibodies to the virus can help us tremendously in our strategy to respond to emergencies, treat patients and care for the elderly.”
China has been doing serology testing for months, and numerous Chinese companies have exported hastily developed kits to countries around the globe. But data from such efforts are not yet published, and accuracy is a problem. The governments of Spain and the United Kingdom have reported buying Chinese tests that didn’t work.
One reason for this is that looking for secondary evidence of a disease-causing germ is tricky. Timing matters; if there aren’t enough antibodies to be detected, that can undermine accuracy. Or, if the test isn’t precise enough in probing for a protein, it may detect antibodies to the wrong virus, a problem called cross-reaction. Other coronaviruses that cause common colds can cross-react with tests for the new virus.
Advaite’s website says, “In testing at a provincial blood bank in China, the Advaite diagnostic test demonstrated accuracy of over 94% in blood samples obtained from COVID-19 infected patients."
Nathan Anderson, whose company, Hindenburg Research, specializes in forensic financial research of biotech companies, said, “Advaite looks like a very small company in a sea of other companies offering these rapid antibody tests. Given the failure rate of these types of tests internationally even from larger manufacturers, it is difficult to get a sense of whether the claimed accuracy is realistic.”
