Pfizer vaccine slashes COVID-19 risk in early trials, sending hopes and stocks soaring
The company said that by the third week of November, it expects to seek FDA authorization to distribute the vaccine on an emergency basis.
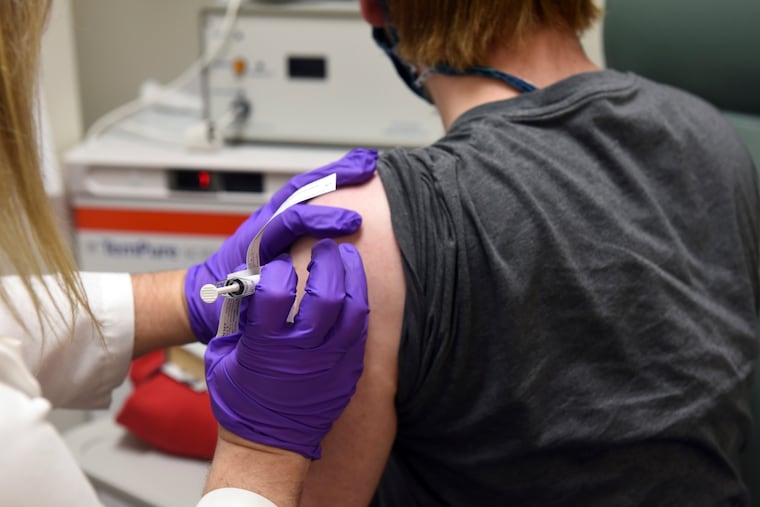
Citing early data, Pfizer Inc. said Monday that its coronavirus vaccine reduces the risk of COVID-19 illness by more than 90%, far exceeding original projections of 60%.
The results were based on 94 confirmed, symptomatic cases of COVID-19 in 39,000 volunteers who had received two doses of either a placebo or the vaccine, which Pfizer developed with German firm BioNTech SE.
That means at least 85 of those people got the placebo (saline injections), whereas nine or fewer cases occurred in people who got the vaccine. No serious safety issues have been identified, the company said.
“With today’s news, we are a significant step closer to providing people around the world with a much-needed breakthrough to help bring an end to this global health crisis,” Albert Bourla, Pfizer chief executive officer, said in a statement.
The company said that by the third week of November, it expected to seek approval from the U.S. Food and Drug Administration to distribute the vaccine on an emergency basis, provided that no safety issues arise before then.
While the numbers are promising, more detail is needed to see how well the vaccine will help prevent severe COVID-19, said infectious-disease specialists who were not involved with the research.
Another hurdle: The drug must be stored at a temperature well below zero, which could mean that widespread distribution will be a challenge. For long-term storage, the required average temperature is negative 94 degrees Fahrenheit, requiring special freezers. But once doses are delivered to a hospital, they can be kept for up to five days at 18 to 28 degrees, well within range of standard freezers, the company says.
The news quickly became political, with Vice President Mike Pence tweeting that President Donald Trump deserved credit for having “forged a public-private partnership” with Pfizer. And some of Trump’s supporters accused the company of withholding the news until after the election, given that Pfizer had previously said interim results might be available in October.
Not so, a senior company official said in an interview with the New York Times. Kathrin Jansen, head of vaccine research and development, said she learned of the results Sunday. As for Pence’s claim, she said Pfizer was “never part of the Warp Speed,” the term for the government’s push to develop a vaccine. It may be a question of semantics: While Pfizer did not receive government funds for research and development of its vaccine, the Trump administration did pledge $1.95 billion to the company in July to support manufacturing and distribution of 100 million doses.
Among researchers expressing caution about the results was Paul A. Offit, director of the Vaccine Education Center at Children’s Hospital of Philadelphia.
“You need to see the data,” he said. “The numbers are small but very encouraging.”
The results were announced only in a press release, and were not broken down by age group, ethnicity, or the presence of any underlying health condition. Such details will be crucial, as older people, people of color, and those with other medical issues are the populations that have been hit hardest throughout the pandemic, Offit said.
In its news release, Pfizer said 42% of participants worldwide and 30% of those in the U.S. have “racially and ethnically diverse backgrounds.”
Despite the lack of detail, the results nevertheless drew optimism from the research community.
“This is really a spectacular number,” Yale University immunologist Akiko Iwasaki told the New York Times. “I wasn’t expecting it to be this high. I was preparing myself for something like 55%.”
Pfizer originally projected that at least 164 trial participants would need to get sick for statisticians to determine whether the vaccine worked, assuming that its true efficacy was 60%. But the interim numbers were so promising that analysts needed just 94 cases to ascertain that the vaccine was preventing illness, and that its apparent potency was not simply due to chance.
Efficacy is used to describe the impact of a drug in the controlled setting of a trial, when researchers can carefully pick how many people get a placebo and how many get the real thing. A vaccine’s effectiveness, on the other hand, is an estimate of how well it works after approval, when anyone can get it.
Nearly 44,000 people have enrolled in the Pfizer trial since it began July 27, though some have yet to receive a second dose. The interim results are based on 39,000 participants who had received two doses as of Nov. 8, the company said.
The Pfizer product relies on a different approach from traditional vaccines, though the end goal is the same: exposing a person to a microbe (or a fragment of it) in such a way that it does not result in illness, so the immune system can learn to fight back in the event of a real infection.
In most traditional vaccines, this exposure is achieved with a weakened or inactivated form of the virus in question. Pfizer’s COVID-19 vaccine, on the other hand, consists of genetic material called RNA, containing instructions for the person’s cells to make a fragment of the coronavirus after it is injected in the body.
A competing COVID-19 vaccine made by Cambridge, Mass.-based Moderna uses a similar approach, though that company has yet to announce results from its phase 3 trial, as Pfizer did Monday.
Yet another type of genetic vaccine for COVID-19 was developed by Inovio Pharmaceuticals of Plymouth Meeting, consisting of DNA, not RNA. But human trials of that product are on hold due in part to FDA questions about the technology used to administer it: a device that delivers electrical pulses to the skin. The Pfizer RNA vaccine, on the other hand, is delivered with a traditional injection.
The news prompted a flurry of investor activity Monday. Pfizer’s stock price increased 7.7% by day’s end, driving gains across much of the market. Inovio’s share price, however, closed down 19%.
No RNA or DNA vaccine has yet been commercialized, though the concepts have been studied for years.
