Breast implant maker's warranty: $7,500 if device causes rare cancer
The first-of-its kind warranty comes as the breast implant industry is being roiled by the mysterious disease, called breast implant-associated anaplastic large cell lymphoma.
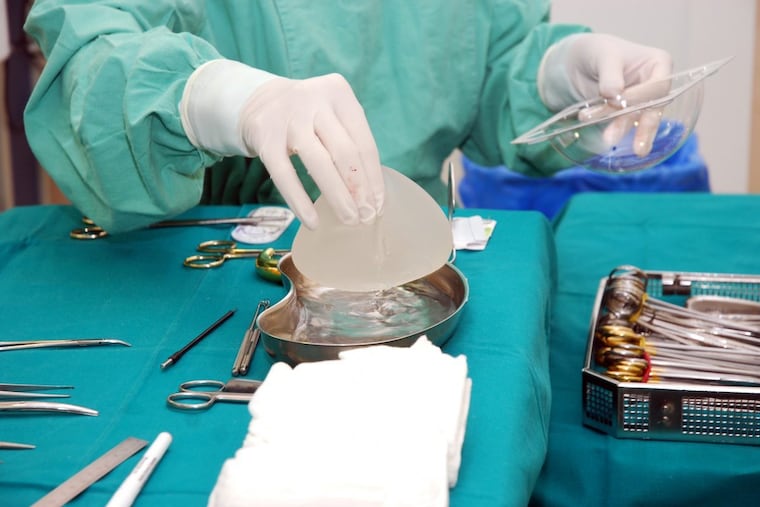
At the urging of the American Society of Plastic Surgeons, Allergan is offering a novel deal to women who choose the company's textured-surface breast implants: $7,500 toward surgical removal if the devices cause a rare immune system cancer.
The first-of-its kind warranty, which the surgeons' society announced this week, comes as the breast implant industry is being roiled by the mysterious disease, called breast implant-associated anaplastic large cell lymphoma (ALCL). In the seven years since the U.S. Food and Drug Administration first warned of the link between textured implants and the lymphoma, it has been diagnosed in 513 women worldwide, including 190 in the United States. Sixteen have died.
Now, attorneys are soliciting patients for lawsuits, disillusioned women are pushing for tougher regulation, and a minority of surgeons are abandoning textured implants – or even saying the products should be taken off the market.
"Most plastic surgeons are of the opinion that ALCL is very rare and as long as we inform patients, we can still use textured implants. I don't agree," said Eric Swanson, a plastic surgeon in the suburbs of Kansas City, Kan. "Even if the risks are small, there are excellent alternatives with smooth implants. I think textured implants should not be sold anymore or be used by plastic surgeons — at least not for cosmetic breast enlargement, which is all I do."
At Penn State Hershey Medical Center, surgeons have sent letters to patients informing them of the risk, their options, and recommending annual check-ups.
"We do not use textured implants anymore," said Penn State plastic surgeon Dino J. Ravnic. "I think it's too early [to stop marketing]. That said, I would not have a woman put them in."
In the U.S., textured devices make up about 13 percent of the implant market. In 2016, implants were used in 290,000 enlargement and 88,000 reconstructive surgeries.
Implant-induced lymphoma — an immune cell malignancy that is not breast cancer — was first recognized in the late 1990s, about five years after manufacturers began using chemicals to etch the surface of some implants. The roughness helps anchor the device in place as tissue grows into the tiny crevices, but it may also trigger chronic inflammation.
Studies have found virtually all implant lymphoma patients have a history of textured implants. The implant filling (saline or silicone gel) and the purpose (breast enlargement or reconstruction after breast cancer) do not seem to matter.
If caught early, the lymphoma can usually be cured with surgery to remove the implants and surrounding scar tissue. However, health insurers don't cover cosmetic implants or related complications, so some cosmetic patients have delayed seeing a doctor about the classic symptom of ALCL — swelling around the implant. Others have had to fight for coverage of lymphoma treatment.
In response to this problem, Mark Clemens, who heads the society of plastic surgeons' committee on ALCL, led discussions with insurers and the three companies that sell implants in the U.S.
"The disease can advance if it's not recognized early and treated appropriately," said Clemens, of MD Anderson Cancer Center in Texas, which has become a leader in ALCL treatment and research. "Now, quite a few insurance companies — not all — have language about covering ALCL."
In addition to financial assistance of up to $7,500 toward out-of-pocket surgical costs, Allergan will provide smooth implants at no charge to patients who opt for replacement.
Clemens called Allergan's updated warranty "a start."
"The expense these women can incur exceeds that amount," Clemens said. "It's important for us to continue working on this."
Neither Allergan nor the other manufacturers, Mentor and Sientra, responded to requests for comment for this article.
But women whose implants have turned them into activists did not hesitate to speak up.
Terri McGregor, 53, of North Bay, Ontario, almost died of metastatic ALCL that caused no symptoms until it was coincidentally diagnosed following her first screening mammogram in 2015. She underwent surgeries, chemotherapies, a targeted drug regimen, a stem cell transplant, and chest radiation at a Toronto hospital. Fortunately, she said, Canada's national health insurance covered all of it.
"Our American women who are diagnosed are desperate if they don't have insurance coverage," McGregor said. "Dr. Clemens would not have invested the effort in this program if there was not a desperate need."
McGregor and Jamee Cook, 40, of the Dallas area, co-administer a two-year-old Facebook page dedicated to informing women about ALCL and helping those who have it. Of 1,469 members, 85 have been diagnosed. Members' posts clearly show that many doctors still have never heard of implant lymphoma.
"At first glance, Allergan's warranty looks like a good deal, but there are several problems with it," said Cook, who suffered a long list of neurological symptoms, but not lymphoma, before she had her implants removed. "Among other things, we worry about waiving rights to litigation if you accept the warranty."
In Cook's view, there is a better remedy: "This is a disease made by these devices. The commonsense thing would be to remove the culprit."
